jflat06 Staff Lv 1
Hey everyone!
This post previews an upcoming Bonus Filter in Foldit design puzzles - the Hydrogen Bond Network Filter.
We are introducing this filter to overcome a major challenge in protein interface design, and something we've observed in many Foldit designs.
A lot of designs we've seen so far have used lots of hydrophobic residues in their interfaces. This works because these hydrophobic residues are buried in the core of the symmetric complex, sandwiched between two copies of the protein.
However, when you are designing proteins with interfaces, you have to consider that placing hydrophobics on the interface means that these residues will be on the surface of each of the individual pieces. This is a problem, because it means that these hydrophobics are exposed in each of the isolated pieces, making each piece unlikely to fold up correctly on its own. If the pieces don't fold up on their own, they wont be around to interface with one another!
To combat this problem, protein designers in the Baker Lab have been using Hydrogen Bond Networks.
A Hydrogen Bond Network is a 'web' of hydrogen bonds that connects the sidechains of multiple residues. When constructed across protein interfaces, these networks help to make the interface more stable. You can see an example of such a network below:

And unlike a hydrophobic core, these networks can be made out of polar hydrophilic residues. Because of this, the network will work well as a surface for each piece - and will still work well as an interface between the pieces!
The other major advantage of Hydrogen Bond Networks is that they increase the specificity of the interface. Since the networks are very carefully joined together like a jigsaw puzzle, each piece will be most happy when it's allowed to network with the other pieces. This helps to ensure that the pieces of your protein will interface with each other like you intended!
Hydrogen Bonds
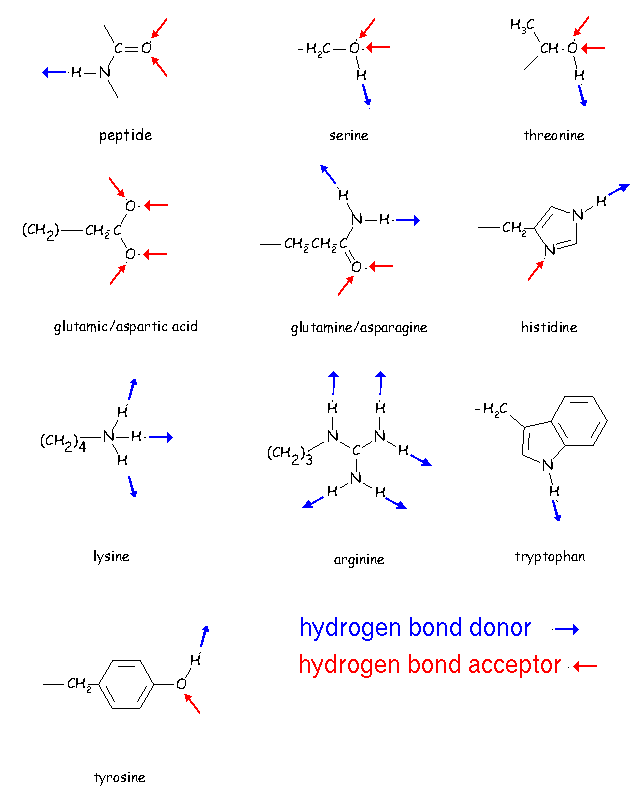
Hydrogen Bonds are an interaction between a donor and an acceptor. As the name suggests, the donor donates a hydrogen atom, and the acceptor accepts it.
In Foldit, you can see these donors and acceptors using certain view options that are available when you've enabled 'Show Advanced GUI' in General Options.
Using the Score/Hydro+CPK Color scheme will color donors blue and acceptors red, like you see in the example above.
Donors have a partial positive charge, and acceptors have a partial negative charge. This causes the donor and acceptor to be attracted. But the opposite charges are not the full story; the charges are in specific locations and directions, and it's the geometry of the h-bond that really dictates its strength.
To get a better idea of how to improve your geometry, you can use the Stick+polarH option in 'View Protein'. This view option will let you see the hydrogen atoms themselves, shown in white.
Hydrogen Bond Networks
When multiple Hydrogen Bonds connect across multiple sidechains, they form a Hydrogen Bond Network. To get credit for a network in Foldit, you must have at least 3 hydrogen bonds, and at least 1 hydrogen bond that connects across an interface (these criteria may change from puzzle to puzzle).
In puzzles where the Hydrogen Bond Network Filter is enabled, you can visualize them using the filter. To do so, open the drop-down Filter Panel beneath the score panel, and click the 'show' checkbox for the HBond Network filter.
Valid networks will show up as a web of blue bonds. Each valid network that you form will get a score, which is then added to your total score as a bonus.
But what makes a good Hydrogen Bond Network?
Well, first off, your hydrogen bonds have to be good! A weak hydrogen bond wont work to extend a network. Remember that the strength of a Hydrogen Bond depends on its geometry. The donor and acceptor must be at the correct distance and angle to be strong enough for a network. Bonds that are too weak will show up in red on the visualization.

Once you have a network of good bonds, the score of a Hydrogen Bond Network is evaluated as follows:
Score = SCORE_SCALE * percent_polars_satisfied * num_intermolecule_bonds
So what do these mean?
SCORE_SCALE - This is just a constant that we set on a per-puzzle basis to decide how much Hbond Networks are worth.
percent_polars_satisfied - A good hydrogen bond network will minimize the number of unsatisfied polar atoms. If there's a blue or red atom in your network, it should be bonded to something - in some cases, multiple times. You want your network to satisfy most or all of its polar atoms.
num_intermolecule_bonds - The more bonds that your network forms across the symmetric interfaces of your protein, the higher it will score!
You can see the stats for each individual network by hovering over one of the bonds of that network. This will also highlight all bonds of that network in yellow:

It's important to note that while Hydrogen Bond Networks are awesome for stabilizing the interface between your symmetric proteins, you should have some hydrophobics on the interface as well. The best solutions will have a balance! Ideally, you want a very connected, perfectly satisfied network, with tight hydrophobic packing around it.
To go along with the release of this filter, we've posted a new puzzle http://fold.it/portal/node/2000714. In this puzzle, the backbone is fixed - your job is to mutate and move the sidechains to form networks of hydrogen bonds. Some players have already created some amazing networks in the devprev version of this puzzle! We're very excited to see what you can do, too!