
beta_helix Staff Lv 1
Voltage-gated ion channels (VGICs) are key elements in cellular electrical signaling, playing crucial roles in physiological processes such as neuron function and muscle contraction.(1) These membrane proteins open or close in response to signals, allowing ions to flow across the cell membrane. Due to their critical role in human physiology, VGICs are important targets for treating conditions like pain, cardiac arrhythmia, and epilepsy. NaV1.7 is particularly significant in pain signaling and is considered a promising target for developing non-addictive painkillers.(2) However, selectively targeting NaV1.7 without affecting other closely related channels has proven challenging due to high sequence and structural similarity within the NaV family.
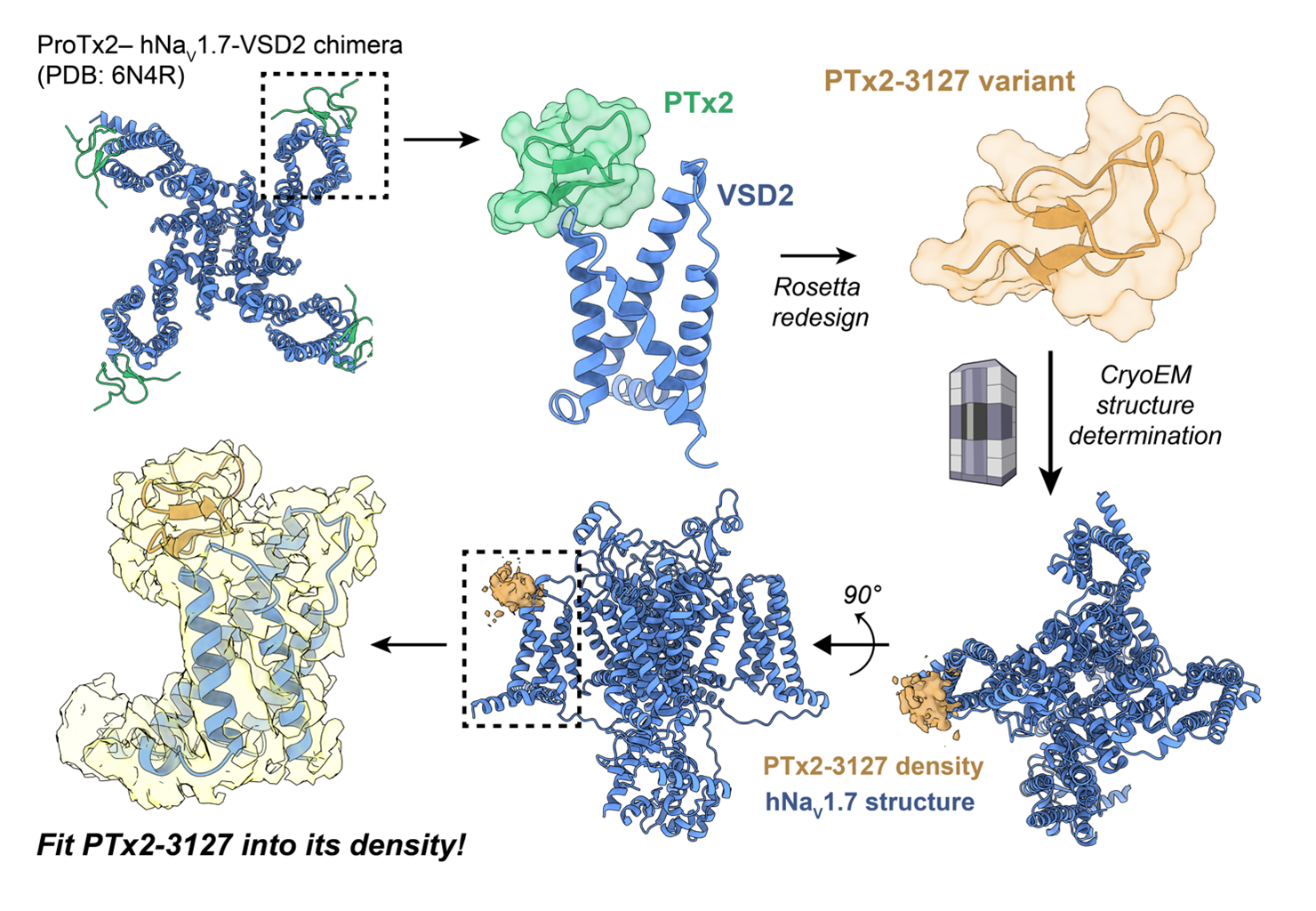
Peptidic toxins derived from animal venoms can modulate VGIC, making them attractive scaffolds for developing therapeutics for these targets.(3) Protoxin-2 (PTx2), a toxin from tarantula venom, is a 30-amino acid peptide that inhibits NaV1.7 by binding to the voltage-sensing domain 2 (VSD2) of the channel and impairing its activation.(4)
In the Yarov-Yarovoy lab, we previously used the experimental structure of PTx2 bound to NaV1.7 VSD2 to conduct Rosetta-based protein redesign.(5) One of the variants, PTx2-3127, exhibited a significantly improved selectivity profile, and could therefore be of potential therapeutic interest. We subsequently aimed to obtain a cryo-electron microscopy (cryoEM) structure of PTx2-3127 bound to the full human NaV1.7 channel by collaborating with Prof. Nieng Yan at Tsinghua University. Although the resulting cryoEM map enabled the construction of an atomic model of the channel, the density corresponding to the toxin variant near VSD2 was not of sufficient resolution to allow us to accurately model the toxin using our current methods. This was the most important part we wanted to see clearly!
With the success of the Foldit community in improving difficult cryo-EM structures, in this Foldit puzzle, we seek your help to create an atomic model of PTx2-3127 bound to the NaV1.7. We provide the experimental EM map and an initial docked placement of PTx2-3127 bound to the VSD2, and ask you to fit the toxin (residues 119-148) into the electron density. The resulting models will help us understand the molecular determinants of PTx2-3127's action on the channel and will inform future design efforts for this important pharmacological target.
(1) Bezanilla, F. Voltage-Gated Ion Channels. IEEE Transactions on NanoBioscience 2005, 4 (1), 34–48. https://doi.org/10.1109/TNB.2004.842463.
(2) McKerrall, S. J.; Sutherlin, D. P. Nav1.7 Inhibitors for the Treatment of Chronic Pain. Bioorganic & Medicinal Chemistry Letters 2018, 28 (19), 3141–3149. https://doi.org/10.1016/j.bmcl.2018.08.007.
(3) Catterall, W. A.; Cestèle, S.; Yarov-Yarovoy, V.; Yu, F. H.; Konoki, K.; Scheuer, T. Voltage-Gated Ion Channels and Gating Modifier Toxins. Toxicon 2007, 49 (2), 124–141. https://doi.org/10.1016/j.toxicon.2006.09.022.
(4) Xu, H.; Li, T.; Rohou, A.; Arthur, C. P.; Tzakoniati, F.; Wong, E.; Estevez, A.; Kugel, C.; Franke, Y.; Chen, J.; Ciferri, C.; Hackos, D. H.; Koth, C. M.; Payandeh, J. Structural Basis of Nav1.7 Inhibition by a Gating-Modifier Spider Toxin. Cell 2019, 176 (4), 702-715.e14. https://doi.org/10.1016/j.cell.2018.12.018.
(5) Nguyen, P. T.; Nguyen, H. M.; Wagner, K. M.; Stewart, R. G.; Singh, V.; Thapa, P.; Chen, Y.-J.; Lillya, M. W.; Ton, A. T.; Kondo, R.; Ghetti, A.; Pennington, M. W.; Hammock, B.; Griffith, T. N.; Sack, J. T.; Wulff, H.; Yarov-Yarovoy, V. Computational Design of Peptides to Target NaV1.7 Channel with High Potency and Selectivity for the Treatment of Pain. Elife 2022, 11, e81727. https://doi.org/10.7554/eLife.81727.
Try out Puzzle 2541: Unsolved Voltage-gated Ion Channel Cryo-EM Density Puzzle here: https://fold.it/puzzles/2013979
