The compound library feature may return molecules that are hard to identify in Foldit. Atoms are identified only by color, and color can be ambiguous at best. It's a accessibility issue for many.
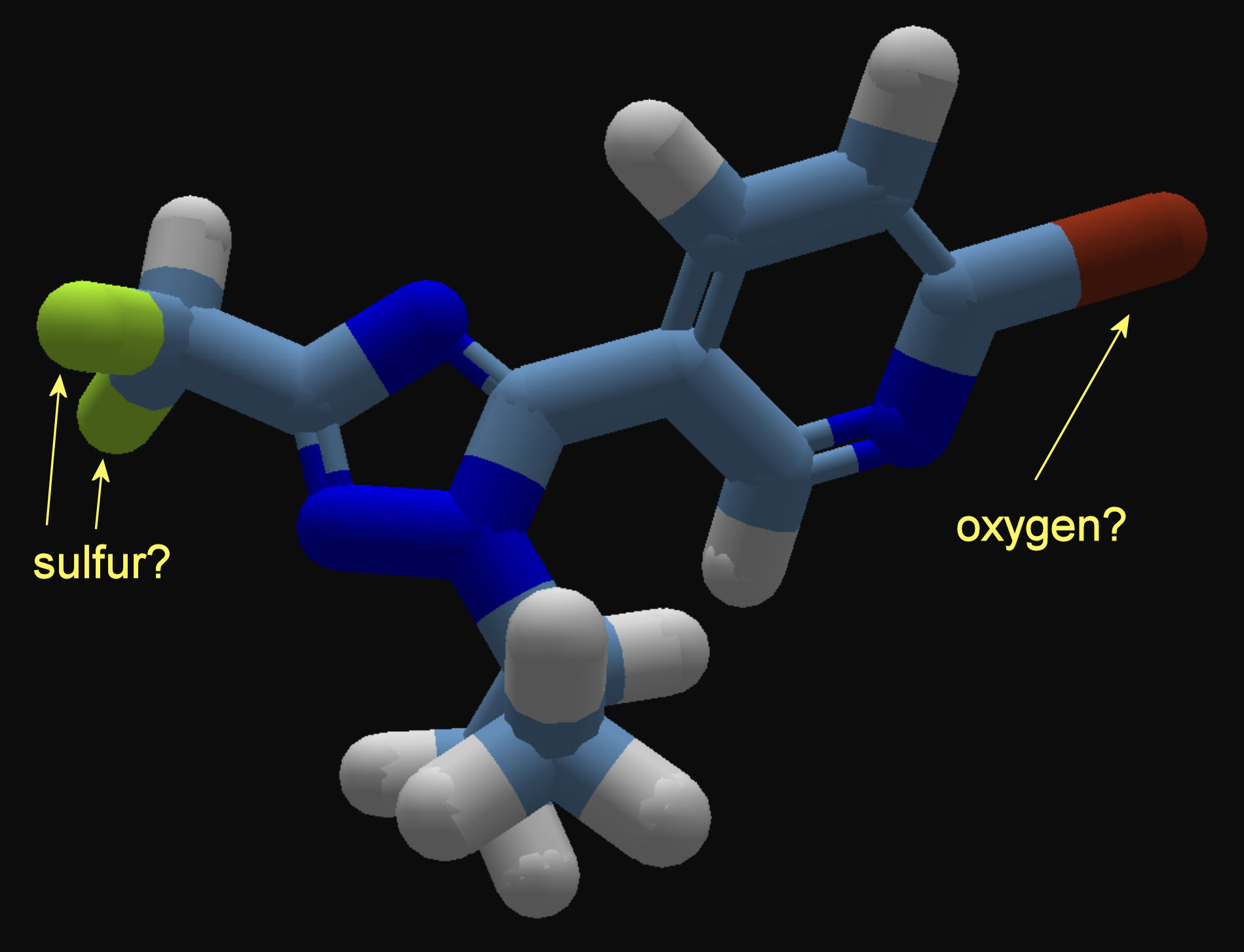
In the molecule shown below, there are two yellowish atoms on the left, and a reddish atom on the right. The yellow color is similar to the one used for sulfur, and the red is similar to the one used for oxygen, but they're a little off.
The "show bondable atoms" and "show all H" view options can help eliminate possibilities. For example, the reddish atom does not show as bondable, so it's not oxygen.
Foldit should have a way to identify individual atoms in the GUI. Hovering over an atom could reveal which element it is.
The should also be a Lua function that returns the element. For example, the function structure.GetElement ( segment, atom ) could return the element for a given atom.
More advanced, but Foldit could also reveal the chemical formula and the SMILES string for a given compound.
The Foldit log.txt does reveal SMILES, so the information is in there. It can be a little tricky to find it, since the log doesn't get written out immediately, and there are no timestamps.
I created this topic in "bugs" by mistake, I meant to post under "suggestions". Unfortunately, there's no way to move it to suggestions, since the "Edit Topic" option is a little too inclusive in terms of destinations. I also don't see a way to delete it and start over.
[Edit: there is an "Edit Topic" button, which allows moving a post to a new forum.]
Following up the SMILES thing, a SMILES string identifies a compound by listing which atoms are connected. Molecule viewers can build a model based on a SMILES string. Foldit uses this format internally, but only reveals it in log.txt messages.
For example, I found this line in log.txt:
interactive.application.actions.ligand_util: {0} Designed Residue: [H]c1nc(Br)c([H])c([H])c1-c1nc(C([H])(F)F)nn1C([H])(C([H])([H])[H])C([H])([H])[H]
It seems to correspond to the molecule in the previous post. The "Br" means the reddish atom is bromine, and the two "F" entries mean fluorine.
I fed the "[H]c1nc(Br)c([H])c([H])c1-c1nc(C([H])(F)F)nn1C([H])(C([H])([H])[H])C([H])([H])[H]" into Jmol, which came up with this view:
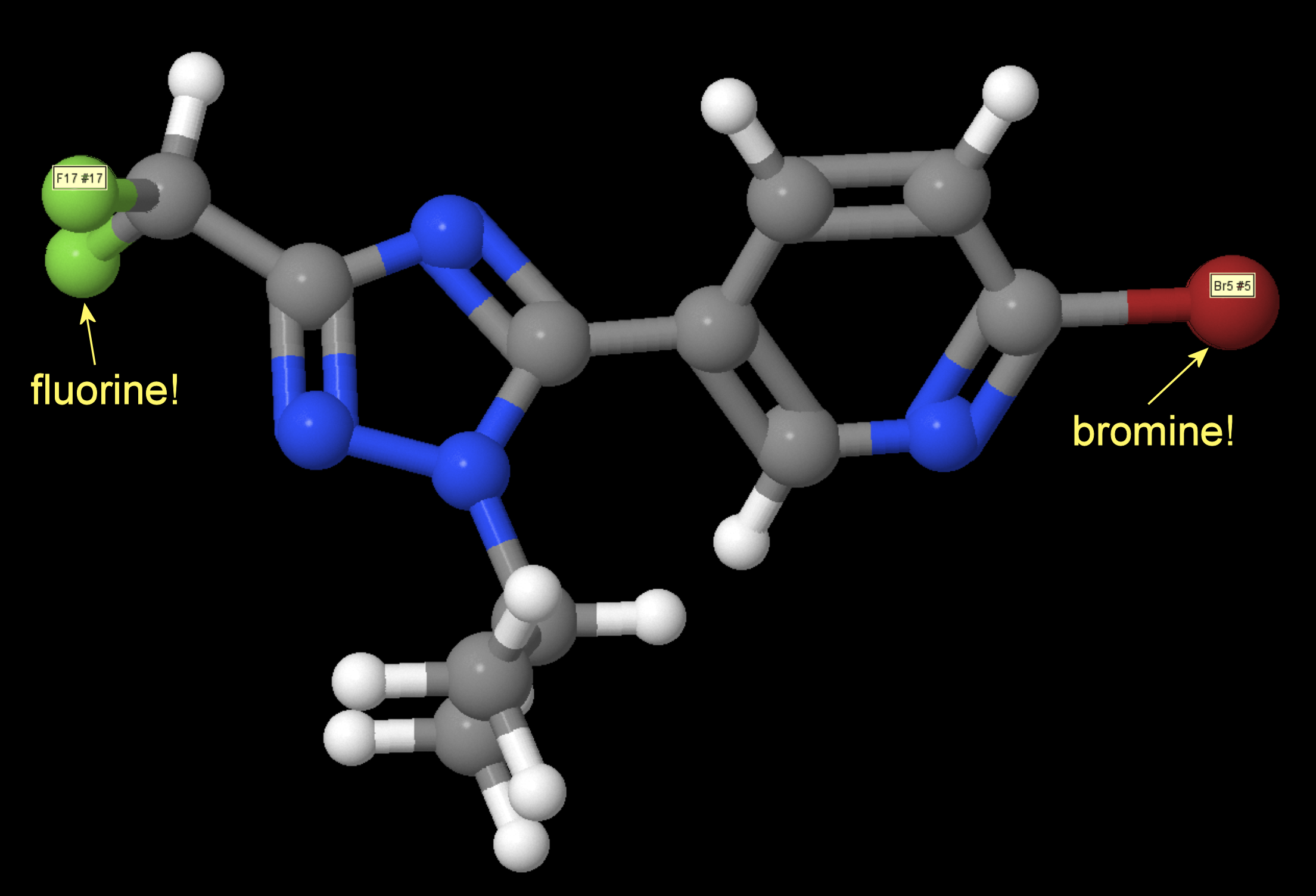
In Jmol, hovering over an atom identifies the element and also the atom number. I've superimposed the identifiers from bromine and fluorine.
Jmol also shows the chemical formula in the title bar, C11H11BrF2N24 in this case. The formula is also shown in the right-click context menu.
Yay, I figured out a way to move this topic to suggestions. The "Edit Topic" link can do it. The link is located above the first post. There's a "Messageboard" dropdown. When you first click the dropdown, you'll hopefully see one of the familiar forums as an option. If you scroll, you'll probably be lost in a forest of forums, which apparently includes an entry for every group ever created, all in no particular order. There's no search option. I'll explore this issue in another post.
That is an informative analysis, LociOiling. It clarified things for me. In the past to the present, I was also hoping for a more expressive view of the ligand in the 2204 and other similar puzzles. One day, I hope to see a WIKI topic about how ligands are represented in the Foldit puzzles.



