
NinjaGreg Lv 1
Two nitrogens that were created by changing a hanging hydrogen to a nitrogen show 2 or three hanging hydrogens (see screenshot). I'll also send the pose to the scientists.
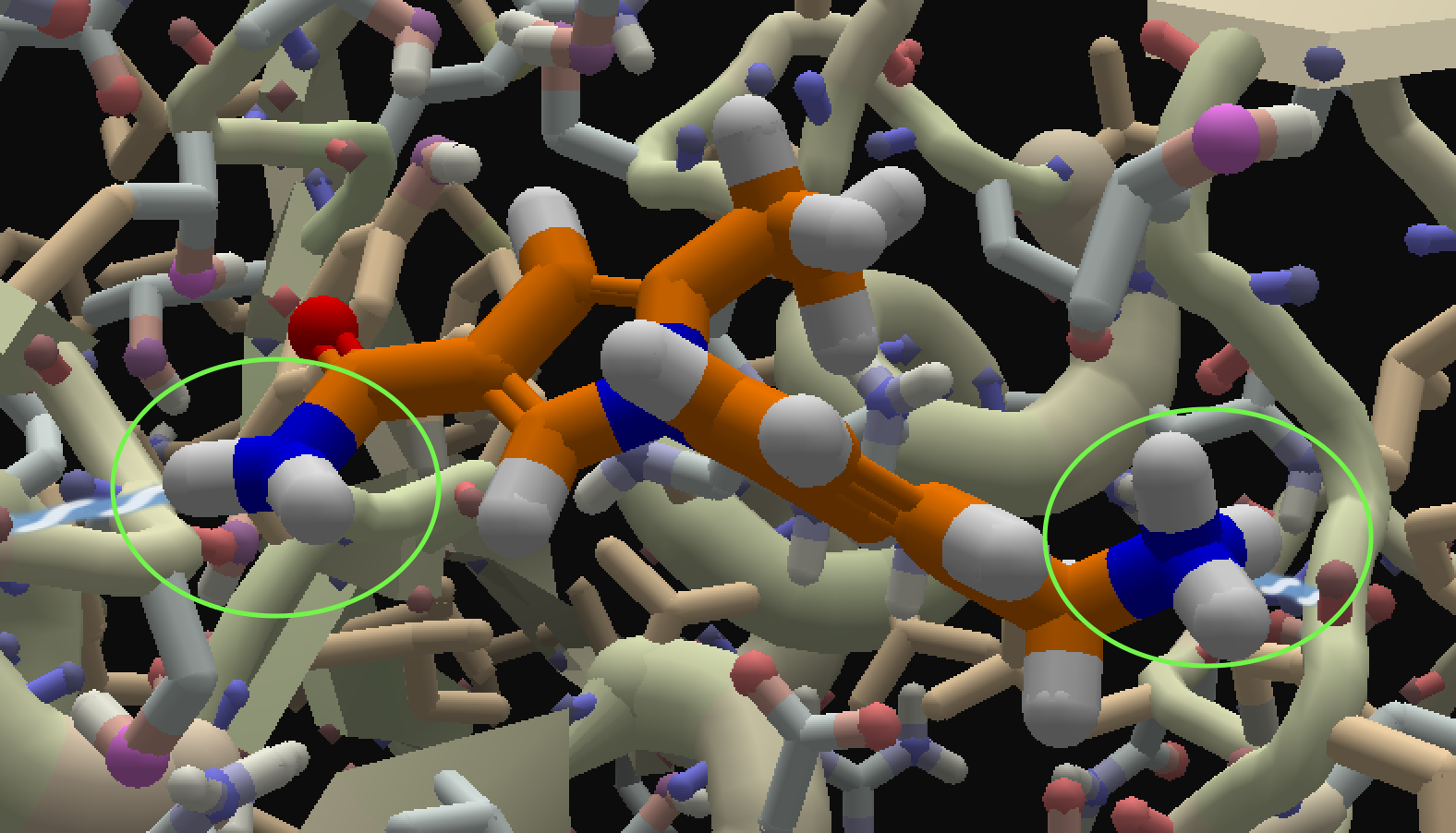

Two nitrogens that were created by changing a hanging hydrogen to a nitrogen show 2 or three hanging hydrogens (see screenshot). I'll also send the pose to the scientists.


This is working as expected. The nitrogen with only two hydrogens is right next to a carbonyl group (C=O), which makes it an amide nitrogen, rather than an amine. With amides, the electron pair that would otherwise pick up another hydrogen is "shared" with the double bond in the carbonyl group, and thus can't bond to the hydrogen. (This is related to the concept of resonance.)
You can actually see this in protein sidechains as well. Compare a lysine sidechain (with an amine) to an asparagine or a glutamine (amides). If you show hydrogens on protein puzzles, you'll see that a lysine has three hydrogens on the sidechain nitrogen, and the asparagine and glutamine sidechains have only two, due to the C=O group which is next to them.

I learn something new every day! My knowledge of chemistry is spartan, at best, so thank you for the explanation!