
Bletchley Park Lv 1
I find myself unable to convert anything into phosphor. Please explain how to do this or consider this a bug report.

I find myself unable to convert anything into phosphor. Please explain how to do this or consider this a bug report.

One way to get phosphorous is by adding a phosphite ion.
Select a hydrogen, then select the phosphite ion from the functional groups area of fragment selection.
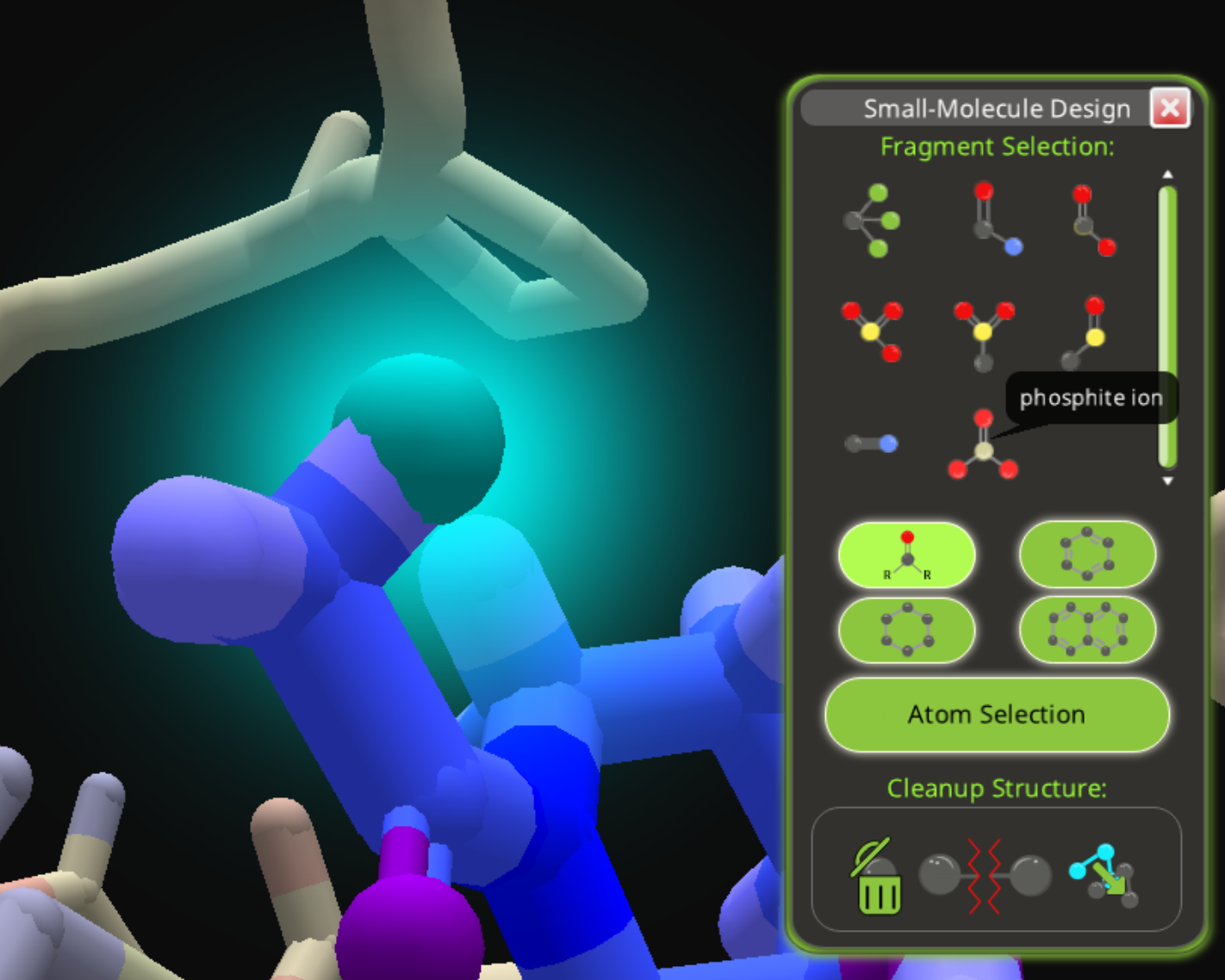
The result is a phosphorous atom bonded to three oxygen atoms. One oxygen has a double bond. There's nothing to positively identify the phosphorous, except the somewhat ambiguous yellow-green color.
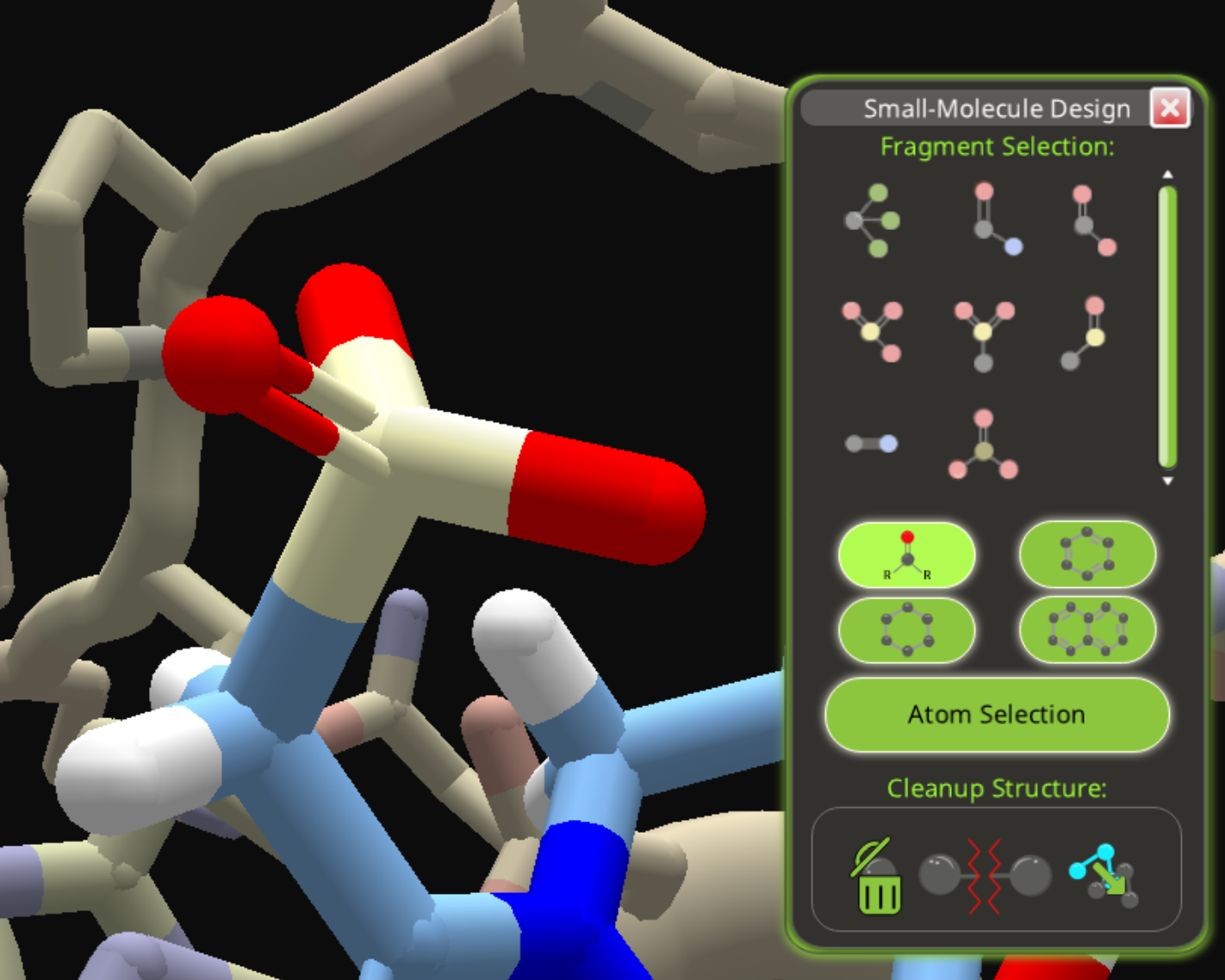
You have limited choices for working with the phosphite. You can change the oxygens to nitrogen or carbon. You won't be able to change the phosphorous or eliminate that double bond.
It's not clear to me whether this is a bug, but it's definitely confusing.

As an alternate path, add a carbon to the same starting spot used in the phosphite example.
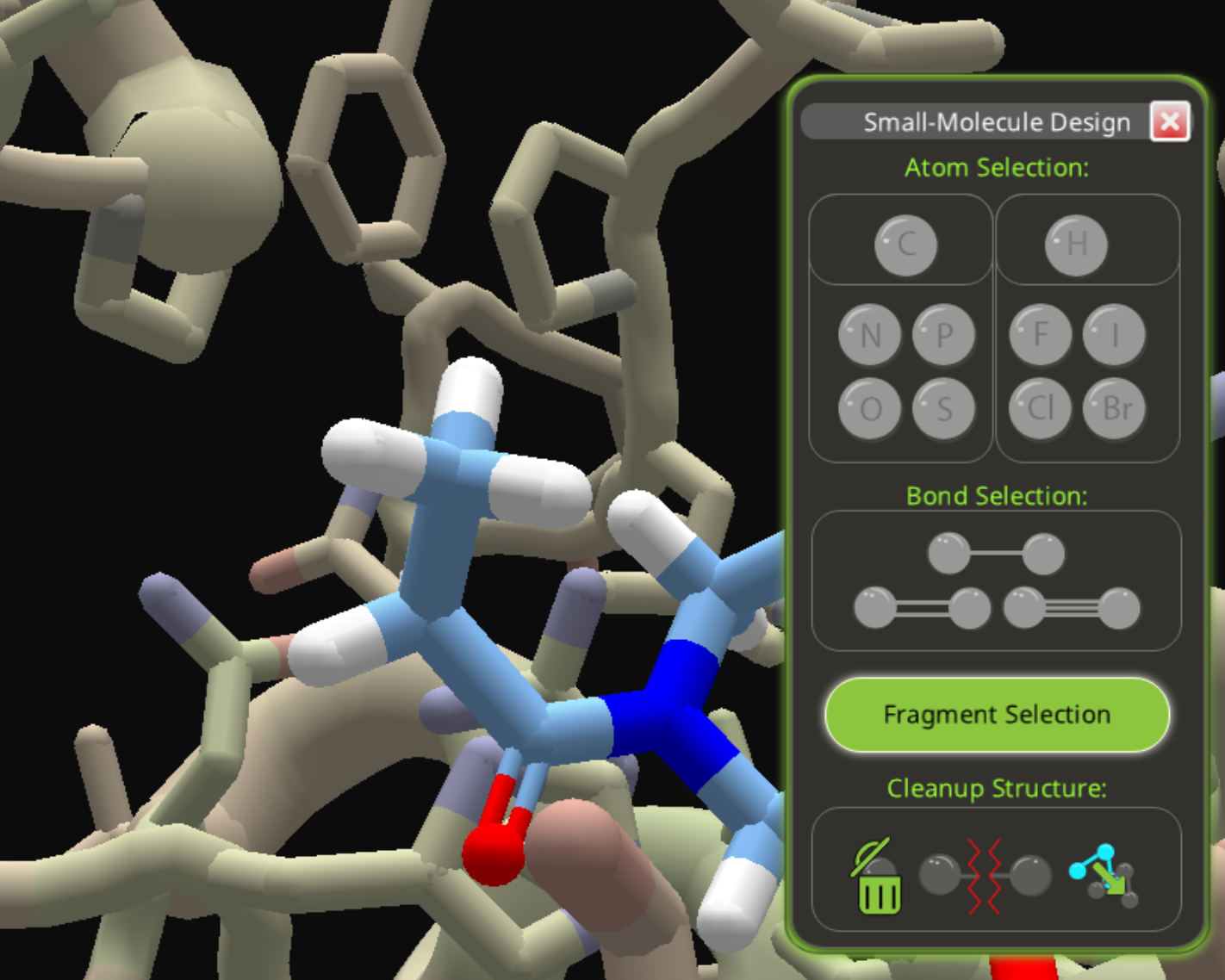
Then add two oxygens to the carbon, replacing two hydrogens.
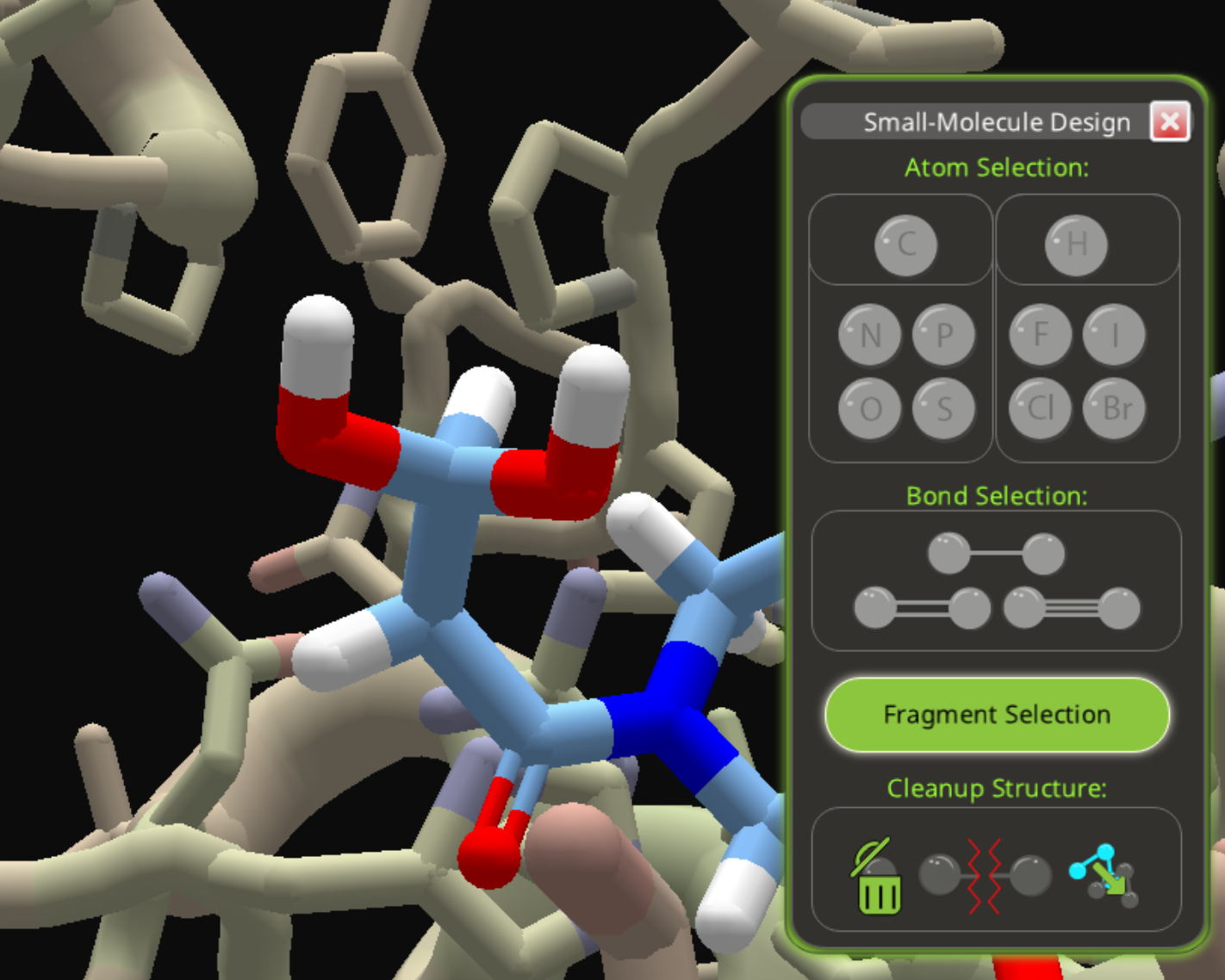
Next, change one of the oxygens to a double bond.
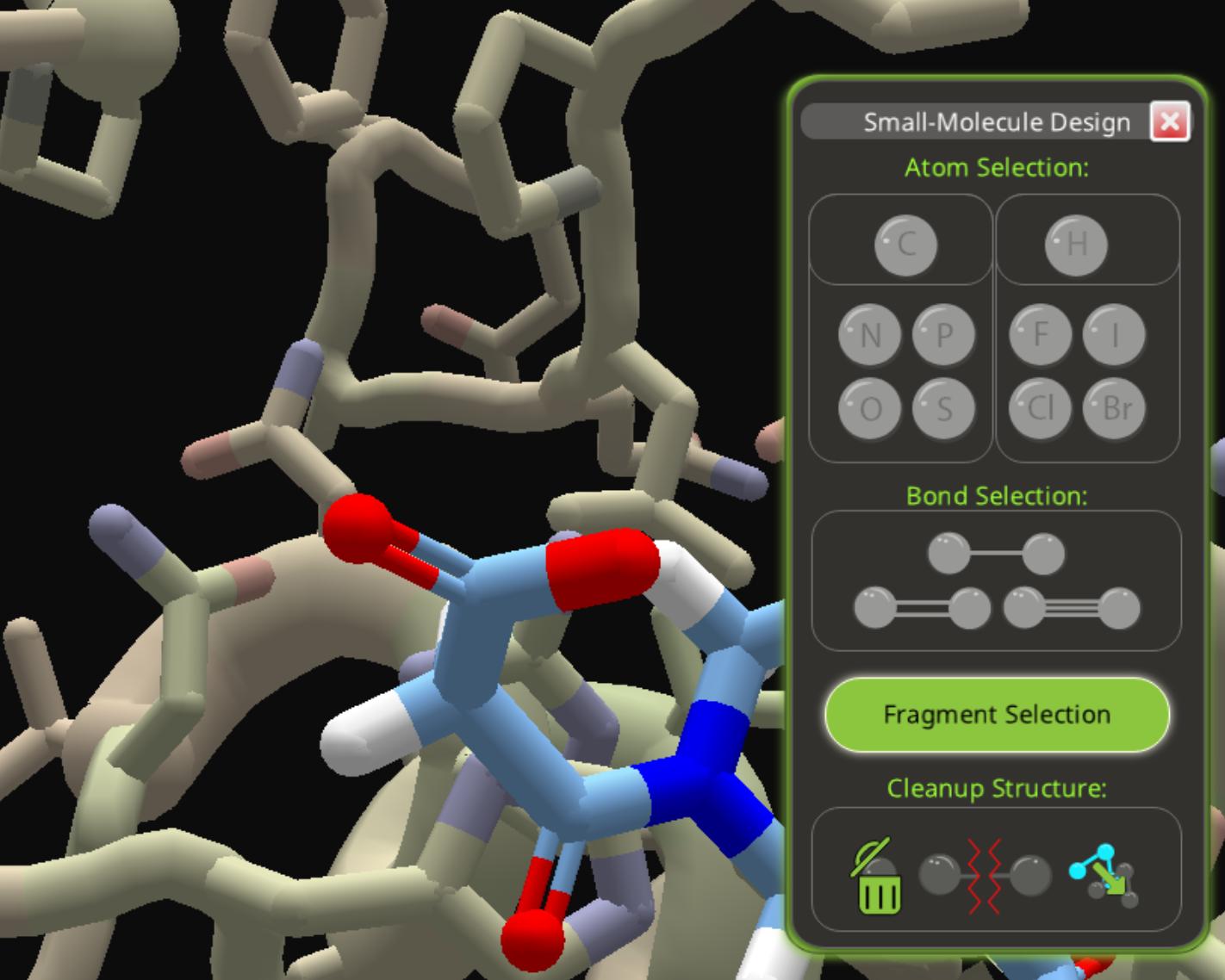
Then replace the carbon with phosphorous. (Edit: left this key step out the first time.)
The result is just one step away from the phosphite in the previous example. There's a phosphorous atom with two oxygens and one carbon. Just change the one carbon to oxygen, and you'll have the phosphite group from the previous example. (Edit: clarify this step.)
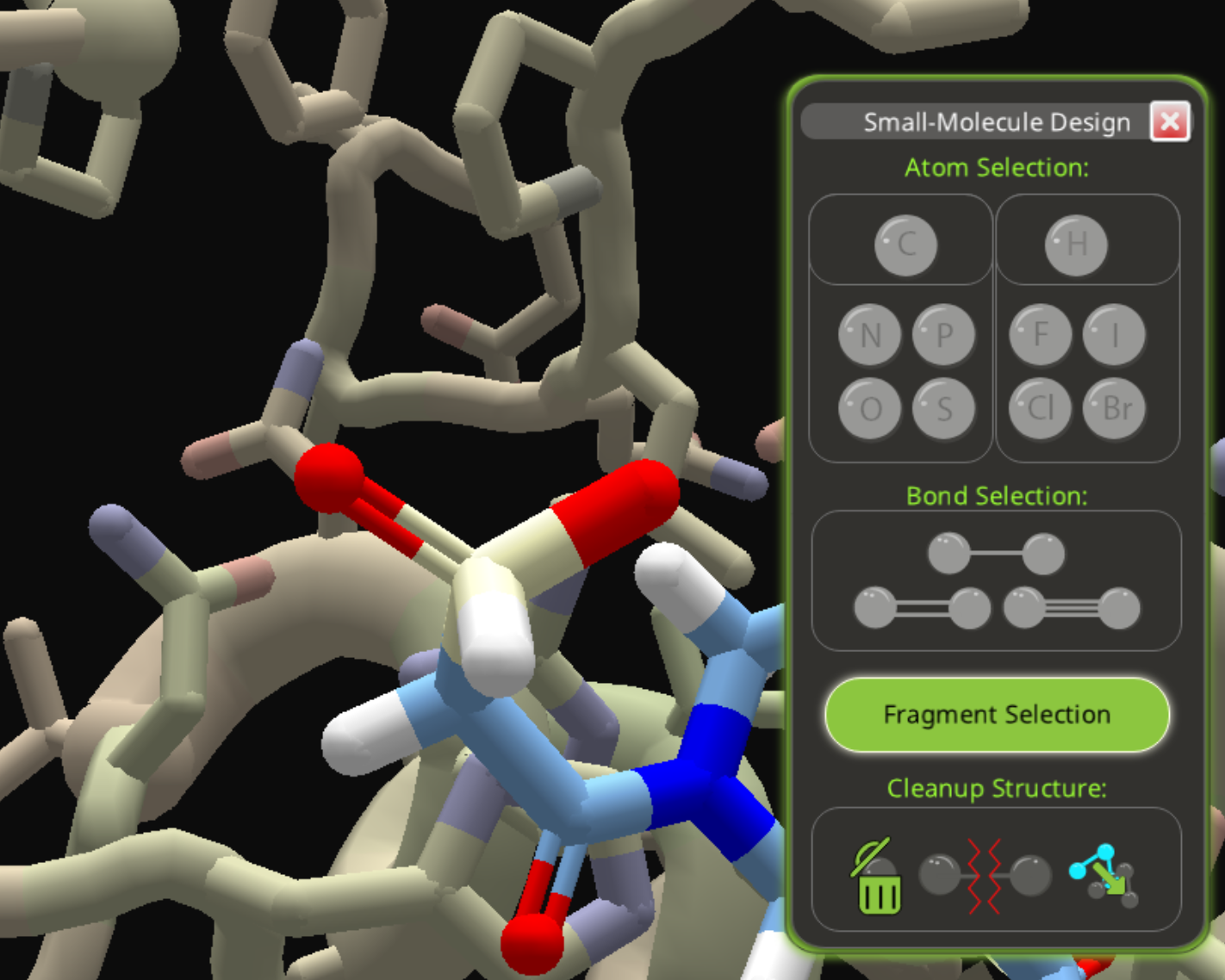
Still confusing, but at least there does seem to be a way to get phosphorous into the mix through substitutions. Getting a library hit may be another matter.

Thank you Loci for explaining this workaround !
In the text you mention "The result is just one step away from the phosphite in the previous example. Just change the one carbon to oxygen, and you're there" I presume, based on the images, that you meant 'change one carbon to Phosphorous' ?
That then also answers my next question to @rmoretti , why is there a P icon in the atom selection if you cannot select P directly. I can imagine that the fact that you need these oxygens to be there first is not well known to the foldit community as it is 'background knowledge' necessary to make this step. N, O and S can be selected directly so to most of us it would seem logical to also select P directly.
Thanks again !

Oops, I left out the key step, changing the carbon to phosphorous. I have edited the post to include that step and clarify what I was saying about the phosphite. In that last image, the atom in the center is already phosphorous, with two oxygens and a carbon around it. Just changing that last carbon to oxygen gets you a phosphite group, as in the previous example.

There's two issues which make changing things to phosphorous an issue. The first is that we're requiring that at every step of the way in modification the compound which you generate is (at least arguably) a valid chemical structure. The other is that phosphorus takes 5 total bonds but only four bound atoms. We normally try to make up the difference in number of bonds by adding/removing hydrogens, but the 5-bonds-to-4-atoms requirement means that you need (exactly) one double bond to the atom you're changing to a phosphorus. If you have that, the change to phosphorous with the atom selection should work. If you don't, then we can't get the order right and you can't add the phosphorus.
It's also the case that phosphorus tends to be rather rare in drug molecules, and typically present only for specialized usages. (Some of out medchem collaborators have argued for removing it entirely.) We've kept it in to allow for those edge cases, but you probably won't be missing anything if you just ignore it. – As Loci says, chances are you won't get a library hit with phosphorus present.

Is it possible to make molecules like ATP or ADP with multiple phosphates connected to each other? If so, please explain how. Is it just a matter of adding one phosphite and then adding another phosphite to the oxygen that is double-bonded to phosphorus? Thanks!

One way to make polyphosphates with the Atom Selection tools is to first make a chain of C & O atoms the length of your polyphosphate chain (with C substituting for P). then add oxygens hanging off the carbons with double bonds (so each carbon is single bonded to each oxygen before/after it and double bonded to an oxygen on the side). Once you have double bonds to your carbons, you can switch out the C for P. This will add an extra H on the P to get the number of bonds right, which you can then substitute out for an oxygen to get your final polyphosphate.

You can even chain things together….
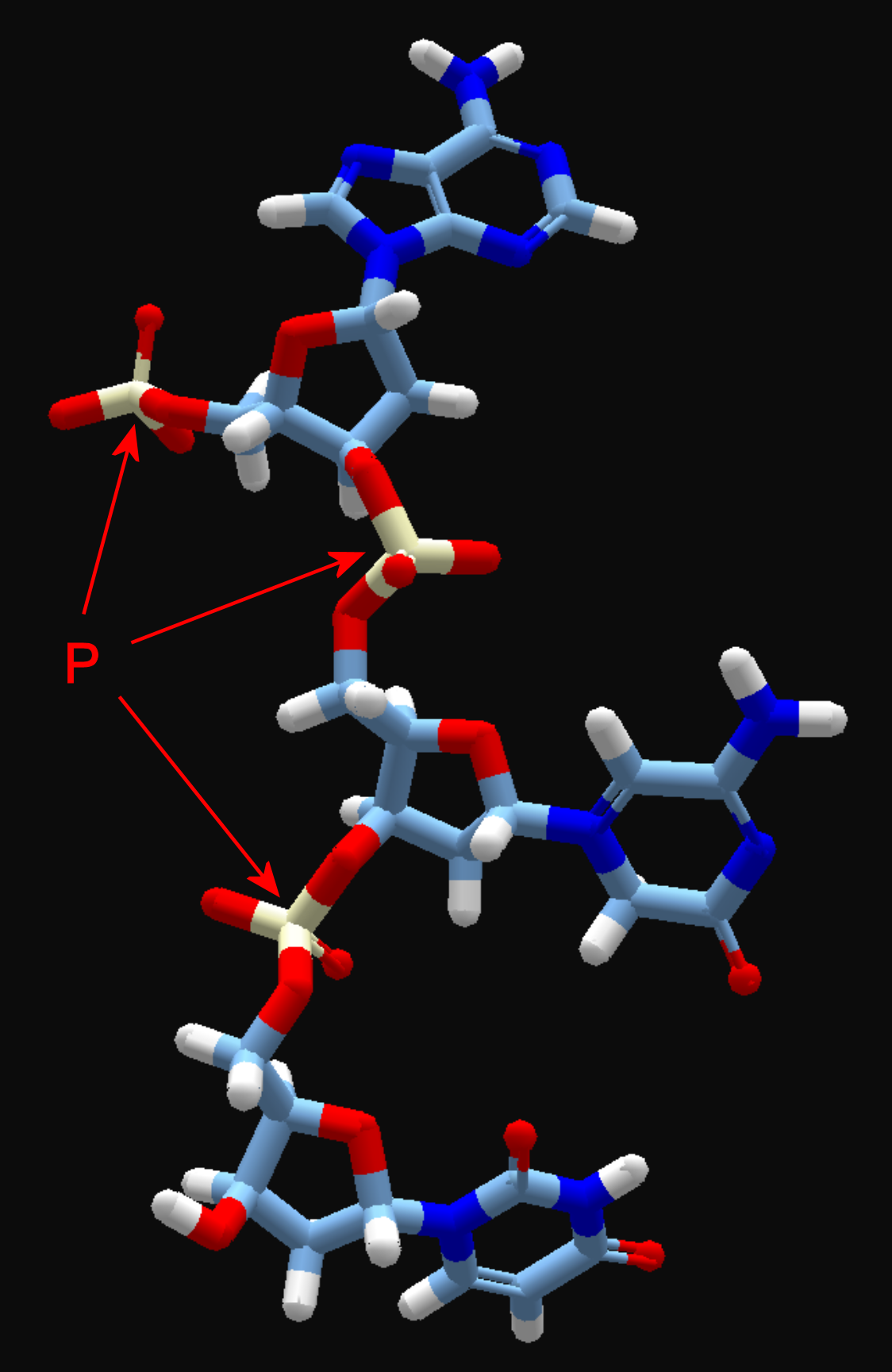
Of course, this example was a ligand TPSA bonus of -10,149….

Thanks to @jeff101 for pointing out the structural flaws in the previous post and providing some detailed advice on how to fix them.
Here's a reworked version, we'll see what the molecule viewer reveals.