dcrwheeler Lv 1
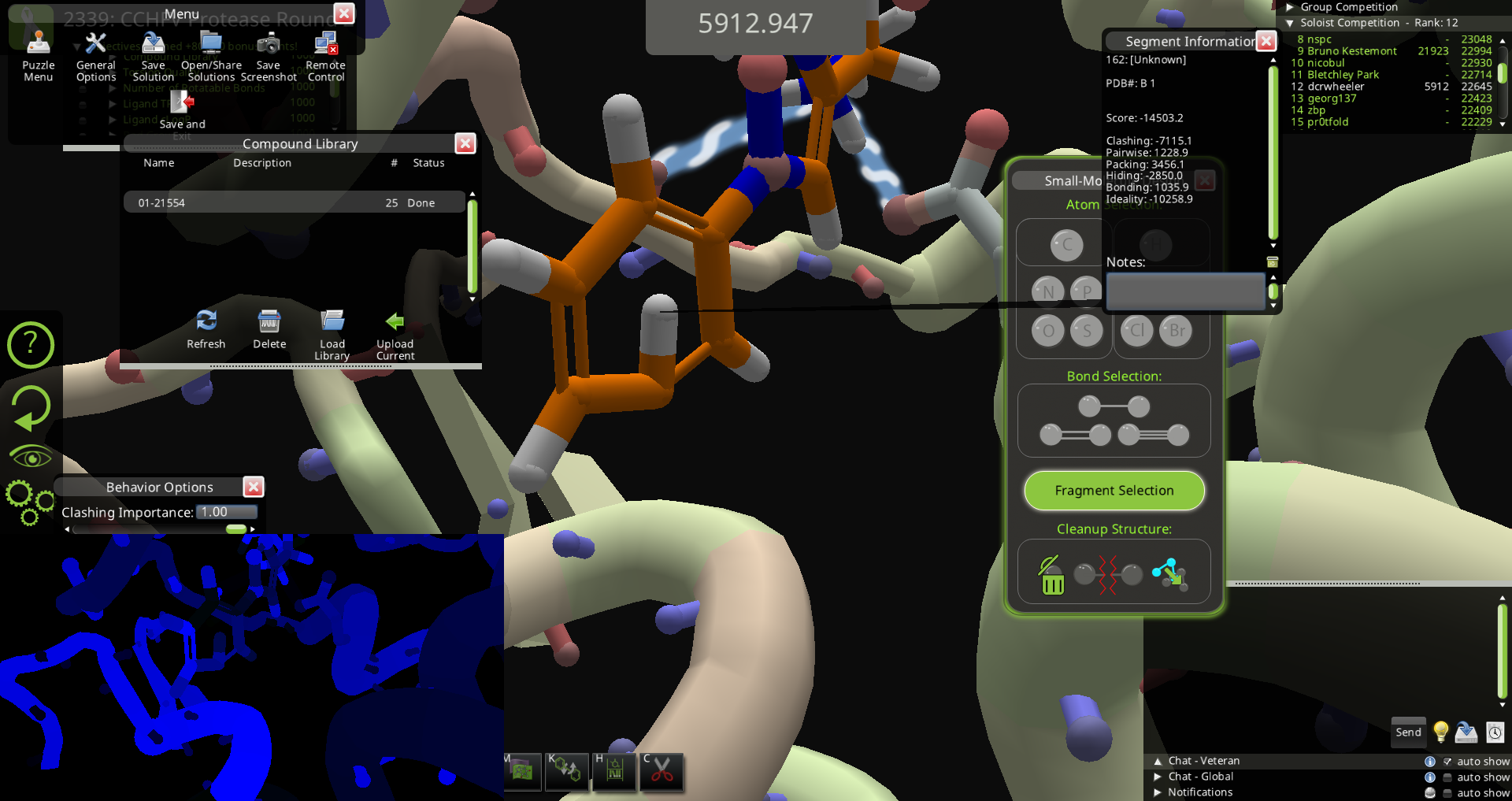
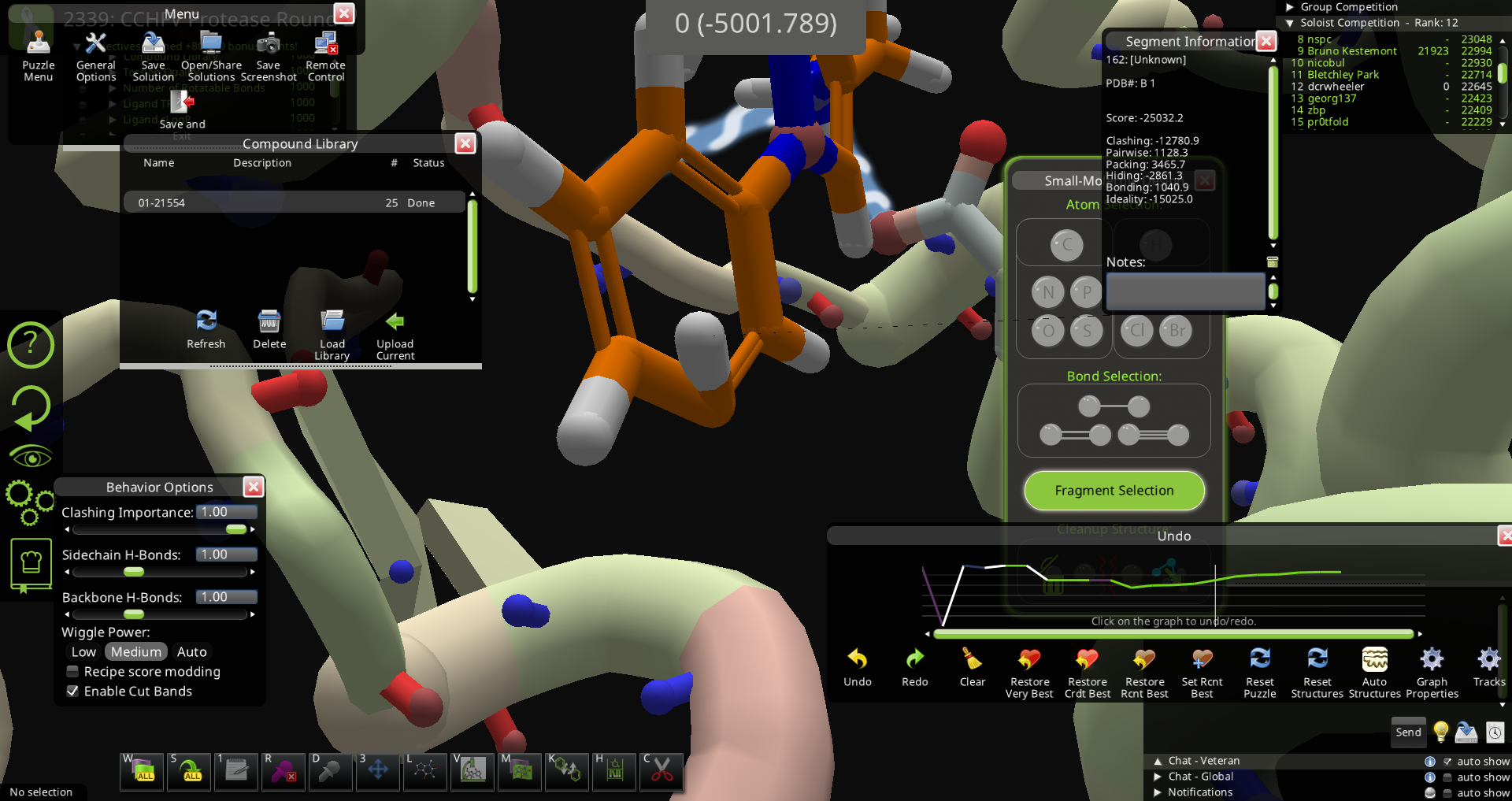



My guess is that it is not realistic. The ideality score is one clue. I'm thinking the H that points to the inside of the phenyl ring is the problem. If you pulled hard on this H with bands at zero clashing importance, you might be able to flip it so it points to the outside of the ring, and then it will give a less negative ideality score. Another way to fix it could be to remove a nearby atom and then rebuild the structure so the H points out of the ring. Perhaps the fix is as simple as running mmff wiggle on it.

This always happen when the ligand has severe clashes with the protein, e.g. overlap with a residue then you wiggle it directly. I've seen worse twists of rings before. Wiggles seems to try removing the overlap but can't fix the structure once it no longer overlap.
As suggested by jeff101 MMFF wiggle can usually fix it quickly.

No, the hydrogen pointing in isn't realistic. As others mention, you can get this if
My attempt at fixing it probably would be to change the adjacent double bond to a single bond and back to a single bond. That should force a rebuild of the hydrogen locations.
(MMFF) wiggling or pulling on the hydrogen could also fix it.