Sciren Staff Lv 1
We are excited to introduce a new challenge, centered around the KCNQ1 gene and its significant role in heart health. KCNQ1 encodes the Kv7.1 voltage-gated potassium ion channel, a crucial player in regulating the heart's rhythm. When mutations occur in this channel, they can lead to serious cardiac conditions, such as congenital long QT syndrome (LQTS). This inherited arrhythmia affects about one in every 2,000 live births and poses a heightened risk of sudden cardiac death, especially in young patients.
Our mission is to find new therapeutics to treat LQTS, focusing on activators of the KCNQ1 channel. One promising compound, ML277, was identified from a large-scale screening of 300,000 compounds. Extensive research revealed key insights into its structure and function:
-
Structural Importance of 4-(4-methoxyphenyl)thiazole: This part of the molecule is crucial for its activity. Any changes to the 4-(4-methoxyphenyl)thiazole part rendered ML277 inactive, meaning it lost its ability to activate the KCNQ1 channel. This highlights the importance of this specific chemical structure in maintaining the compound's function.
-
Chirality and Activity: ML277 has two forms, known as isomers, which are like mirror images of each other. The (R)-isomer is the active form that effectively activates the KCNQ1 channel. The other form, the (S)-isomer, does not have this activating effect. This demonstrates the significance of the molecule's 3D shape in its function.
-
Role of the Tolyl Group: The tolyl group, a specific part of ML277, was identified as the most effective part of the molecule. This group interacts favorably with the KCNQ1 channel, contributing significantly to the compound's ability to activate the channel.
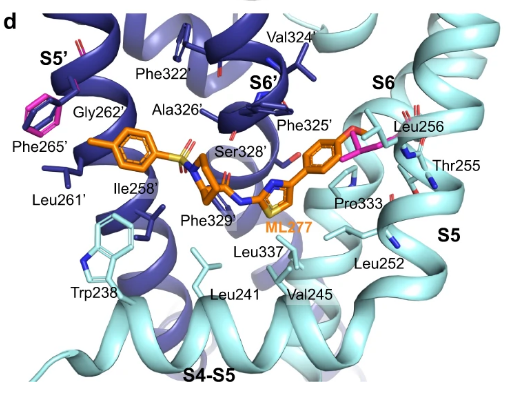
Recent CryoEM studies have provided a detailed view of how ML277 interacts with KCNQ1, helping us understand its role as an activator. ML277 fits snugly alongside a specific helix linker in the KCNQ1 channel, causing it to open and restore proper function. It will be a good idea to keep these areas in mind as both the thiazole and the tolyl group create hydrophobic interactions with KCNQ1. That being said, it is not explicitly necessary to preserve these as exploration in the chemical space is important.
.png)
- Enlarged side view of the ML277 binding pocket formed by the S4-S5 linker helix.
- Willegems, K., Eldstrom, J., Kyriakis, E. et al. Structural and electrophysiological basis for the modulation of KCNQ1 channel currents by ML277. Nat Commun 13, 3760 (2022). https://doi.org/10.1038/s41467-022-31526-7
For this puzzle, our goal is to design a new KCNQ1 activator that can restore function in KCNQ1 variants associated with LQTS. We started with a virtual screening of a massive library containing 36 billion compounds and narrowed it down to less than 900 candidates. These compounds were selected for their potential to interact effectively with KCNQ1 at the ML277 binding site. Since extensive research has been done to optimize ML277, we will be using some of these other candidates as starting molecules in this puzzle series to help broaden the search and development efforts.
Now, it's your turn to join the quest! Since scientists are still working on a therapeutic intervention for LQTS, and their primary guide, currently is ML277, it is important to help their efforts by finding other promising compounds that bind with KCNQ1. Help us discover a new activator that could make a real difference in the lives of those affected by LQTS. Let's work together to unlock the potential of KCNQ1 and bring new hope to those with this challenging condition!

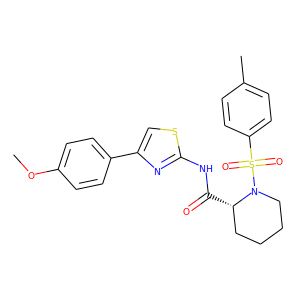 Below is the image the tocris site gave:
Below is the image the tocris site gave: