In Round 1, I stumbled upon a compound that was almost symmetrical.
After one or two modifications it was completely symmetrical; a molecular version of a palindrome: 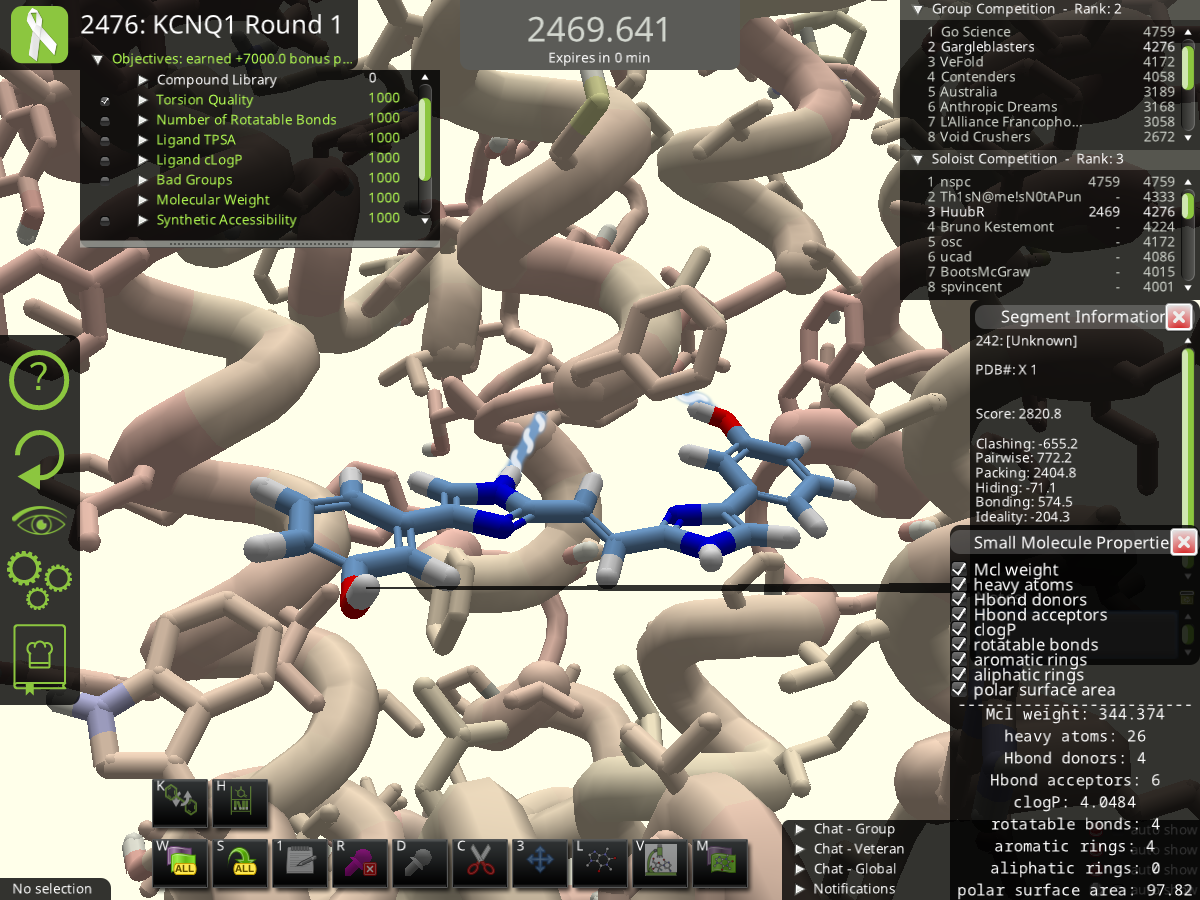 (Shared with Scientists last Monday, 01 July 2024, Puzzle 2476, at 2453 points).
(Shared with Scientists last Monday, 01 July 2024, Puzzle 2476, at 2453 points).
That made me wonder: would this symmetry have an effect on the binding properties? Just like it's easier to insert a key into a lock when the key has identical dents on both sides, or to plug a USB-C connector into its socket, I can imagine that a symmetrical molecule has a better chance of finding its most favourable orientation in the binding pocket.
So my question is: does symmetry have any effect, or is it totally irrelevant?
The binding affinity of a ligand to the protein has both enthalpic (energetic) and entropic (ordering) components. I haven't done the full statistical mechanics calculations, but I would imagine there's a slight entropic bonus for a symmetric molecule binding to a protein. (You lose less entropy going from an unbound molecule tumbling randomly in solution to a molecule fixed in the protein, as there's theoretically more ways to be bound.) – That effect is going to be minor, though, and is likely to be swamped by the ability of an asymmetric molecule to make better, more specific interactions with the protein. (i.e. what you lose in entropy you more than make up in potential enthalpy gains.)
The one big benefit of symmetric molecules would actually be in synthesis - most drugs are built by "convergent synthesis", where you take smaller independent parts and stick them together. For a symmetric molecule, this process is made easier as you can synthesize the two halves and stick them together. (Additionally, any post-combination reactions can proceed on both halves identically.) Though this isn't necessarily all that great of benefit. Normally getting a better-performing compound is more than worth the extra pain of a more complicated synthesis.
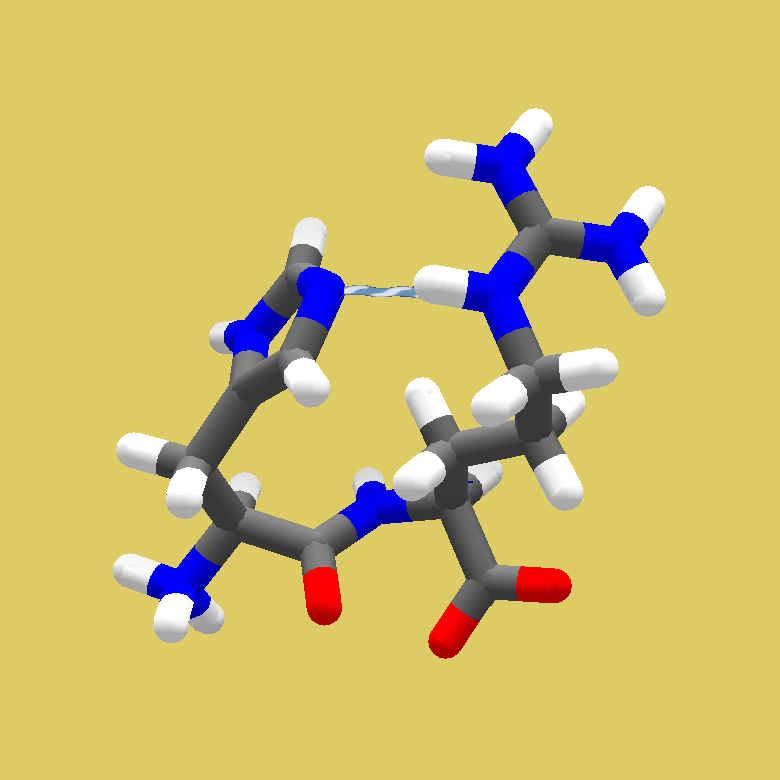
 (Shared with Scientists last Monday, 01 July 2024, Puzzle 2476, at 2453 points).
(Shared with Scientists last Monday, 01 July 2024, Puzzle 2476, at 2453 points).